Kybella (Dexycholic Acid) Injection For Submental Fullness
Disclaimer: Results may vary patient to patient. There is no guarantee that any specific result can be achieved. Results may not be permanent.
Ask your SOMA Skin & Laser dermatologist if Kybella is right for you. Call now for a consultation: 973-763-7546.
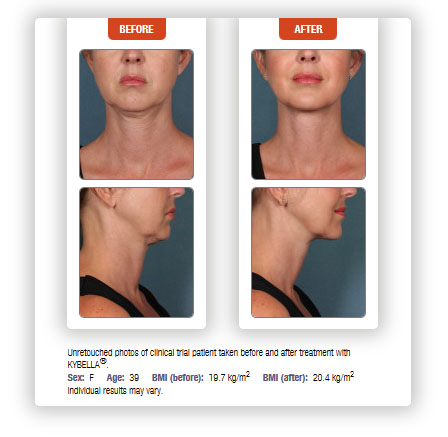
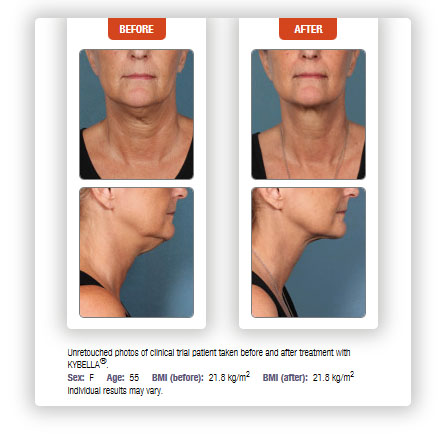
If you suffer from fullness under your chin, sometimes called a double-chin, or more medically submental fullness, Kybella might be the answer. Kybella is an injectable treatment that can dissolve small areas of unwanted fat, and is approved for the treatment of submental fullness.
Submental fullness is a very common problem, with up to 67% of consumers troubled by it, according to a survey by the American Society for Dermatologic Surgery (ASDS).
How often is Kybella injected?
Kybella injections are performed monthly for up to six months, but many patients experienced visible results in two to four treatment sessions. More than six treatments can be performed if needed. Sessions should be spaced at least one month apart. In the Kybella clinical trials, up to six session were administered at least one month apart. Nearly 70% of patients experienced a ≥1-grade improvement with Kybella.
What is Kybella?
Kybella is a brand name for a formulation of deoxycholic acid. Kybella is an injectable medication used for submental fullness (double chin)
What is deoxycholic acid?
Deoxycholic acid (brand name Kybella) is able to dissolve fat. It is also naturally found in the the human digestive system. When Kybella is injected into subcutaneous fat, the fat cells are destroyed. Once destroyed, those cells cannot store or accumulate fat.
What happens at a Kybella treatment?
At a Kybella treatment session, your dermatologist will mark the area to be injected and determine how many injections and how much Kybella is required. The injections are done with a fine needle and are well-tolerated. Kybella is administered by subcutaneous injections directly into submental fat via injections spaced 1 cm apart using a small needle. In clinical trials, the average dose was typically 2-3 vials (4-6 mL) per patient treatment session. However, in our office setting, 1-2 vials is more typical. In-office treatment sessions are typically 15-20 minutes long. At follow-up visits, your doctor will determine the need for further treatments.
Disclaimer: Results may vary patient to patient. There is no guarantee that any specific result can be achieved. Results may not be permanent.
How effective is Kybella in reducing submental fat?
According to clinical studies, 68.2% of subjects treated with Kybella experienced a ≥1-grade improvement with Kybella compared to 20.5% of placebo-treated subjects, based upon validated physician and patient measurements.
What are the side-effects and risks from Kybella?
The most commonly reported adverse reactions clinical trials were: injection site edema/swelling, hematoma/bruising, pain, numbness, erythema (redness), and induration (hardness). Pain, swelling and bruising are likely to occur. In clinical trials, 72% of subjects treated with Kybella experienced hematoma/bruising.
Any injection, including Kybella, can result in an infection. Skin should be cleaned thoroughly before injection. Makeup should not be worn in that area on the day of the procedure.
Cases of marginal mandibular nerve injury, manifested as an asymmetric smile or facial muscle weakness, were reported in 4% of subjects in the clinical trials. All cases resolved spontaneously (range 1-298 days, median 44 days). Kybella should not be injected near a facial nerve.
Dysphagia (difficulty swallowing) occurred in 2% of subjects in the clinical trials in the setting of administration site reactions, e.g., pain, swelling, and induration of the submental area; all cases of dysphagia spontaneously resolved (range 1-81 days, median 3 days).
Kybella should be used with caution in patients with bleeding abnormalities or who are currently being treated with antiplatelet or anticoagulant therapy as excessive bleeding or bruising in the treatment area may occur.
To avoid the potential of tissue damage, Kybella should not be injected into or in close proximity (1-1.5 cm) to salivary glands, lymph nodes and muscles.
Can Kybella be used to dissolve fat in other places?
Kybella is only approved for use in the submental area, but in off-label usage, other areas with fat have been treated as well, including lipomas. Kybella can also be used to treat under eye fat pads.
How much does Kybella cost?
Contact SOMA Skin & Laser to get a Kybella consultation, and customized pricing.

